Indium is a chemical element with symbol In and atomic number 49. It is a post-transition metallic element that is rare in Earth's crust. The metal is very soft, malleable and easily fusible, with a melting point higher than sodium, but lower than lithium or tin.

- Element Indium (In), Group 13, Atomic Number 49, p-block, Mass 114.818. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images.
- Indium (atomic number 49, symbol In) is a malleable and soft chemical element, which is similar to thallium and gallium in terms of chemical properties. It is a rare and fusible metal and exhibits intermediary properties between gallium and thallium. The primary source of indium is zinc ores, and it occurs in a compound form in them.

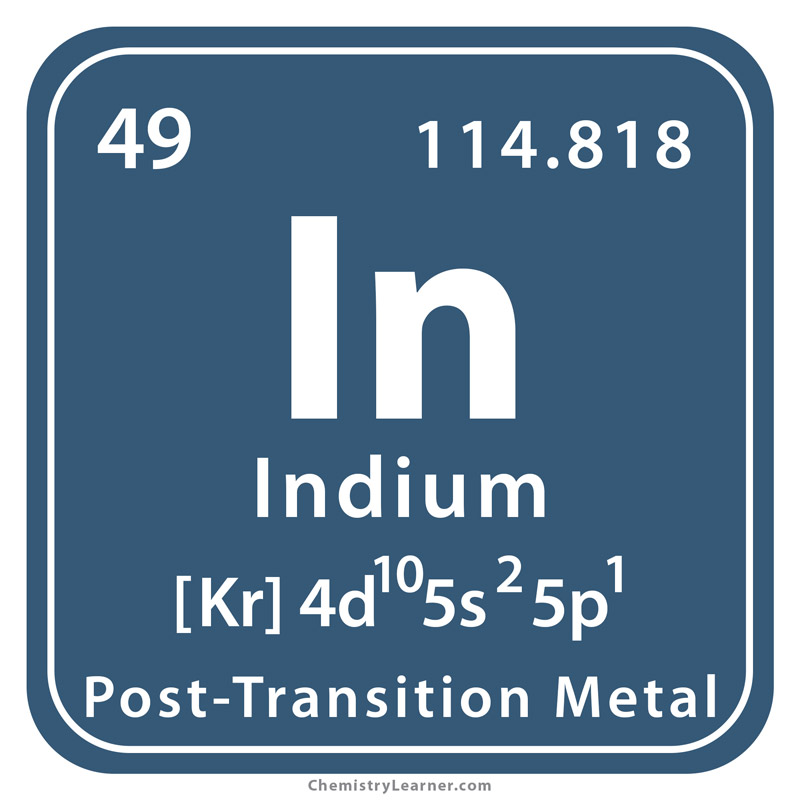
Atomic Number of Indium is 49.
Chemical symbol for Indium is In. Number of protons in Indium is 49. Atomic weight of Indium is 114.818 u or g/mol. Melting point of Indium is 156,2 °C and its the boiling point is 2080 °C.
» Boiling Point» Melting Point» Abundant» State at STP» Discovery YearAbout Indium
Indium is a soft light blue and silver color metal, with chemical properties similar to the ones of gallium or sodium. Uncombined indium almost does not exist on our planet, but it is possible to obtain it from minerals and natural ores. The name of this chemical element is derived from the Latin word meaning indigo. This chemical element has no biological importance and can be even dangerous to living creatures, even in very small doses. Indium creates a number of compounds like oxidants and other organic compounds. Indium is used in alloys and for producing a large numbers of goods like solar panels, LED and touch screens, etc. Combined with other chemical elements, indium is used in thermometers, diodes, dental amalgam, transistors, microchips, and so on.

Uses of Indium
Compounds with Indium
- ITO: Indium tin oxide
- InN: Indium nitride
- In2O3: Indium(III) oxide
- InSb: Indium antimonide
- InP: Indium phosphide
- TMI: Trimethylindium
- InGaP: Indium gallium phosphide
- InGaN: Indium gallium nitride
- InF3: Indium trifluoride
- InAs: Indium arsenide
- CIGS: Copper indium gallium selenide
Properties of Indium Element
| Atomic Number (Z) | 49 |
|---|---|
| Atomic Symbol | In |
| Group | 13 |
| Period | 5 |
| Atomic Weight | 114.818 u |
| Density | 7.31 g/cm3 |
| Melting Point (K) | 429.75 K |
| Melting Point (℃) | 156,2 °C |
| Boiling Point (K) | 2345 K |
| Boiling Point (℃) | 2080 °C |
| Heat Capacity | 0.233 J/g · K |
| Abundance | 0.25 mg/kg |
| State at STP | Solid |
| Occurrence | Primordial |
| Description | Metal |
| Electronegativity (Pauling) χ | 1.78 |
| Ionization Energy (eV) | 5.78636 |
| Atomic Radius | 155pm |
| Covalent Radius | 144pm |
| Van der Waals Radius | 193 |
| Valence Electrons | 3 |
| Year of Discovery | 1863 |
| Discoverer | Reich and Richter |
What is the Boiling Point of Indium?
Indium boiling point is 2080 °C. Boiling point of Indium in Kelvin is 2345 K.
What is the Melting Point of Indium?
Indium melting point is 156,2 °C. Melting point of Indium in Kelvin is 429.75 K.
Indium Atomic Number And Mass
How Abundant is Indium?
Abundant value of Indium is 0.25 mg/kg.
What is the State of Indium at Standard Temperature and Pressure (STP)?
INDIUM - Pomona
State of Indium is Solid at standard temperature and pressure at 0℃ and one atmosphere pressure.
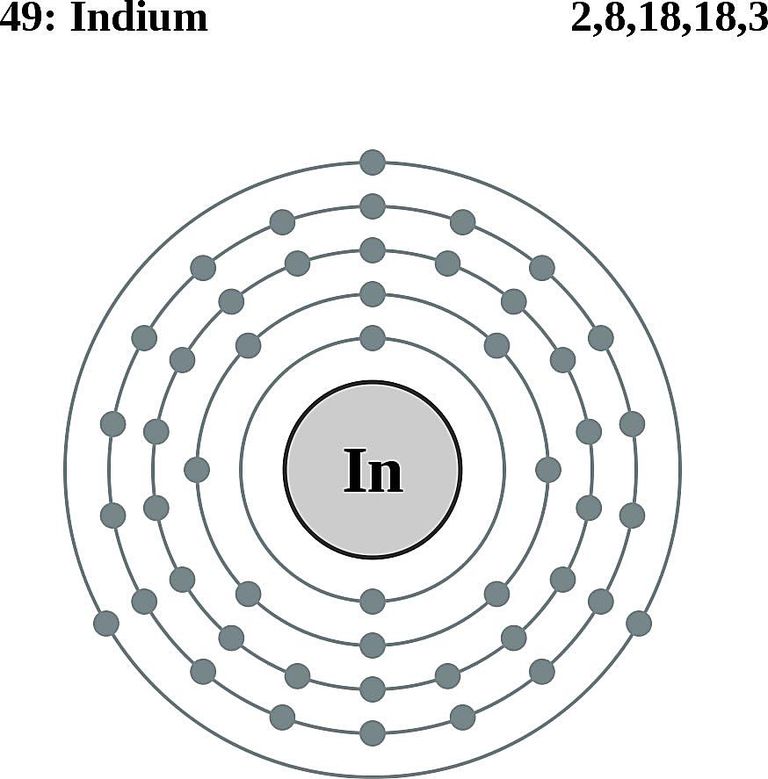
Indium Electrons
When was Indium Discovered?
Atomic Number - Indium
Indium was discovered in 1863.
Indium Bohr Model
