Element Fluorine - F
- It has 5 valence electrons in the 2p level. Its electron configuration is 1s22s22p5. It will usually form the anion F- since it is extremely electronegative and a strong oxidizing agent. Fluorine is a Lewis acid in weak acid, which means that it accepts electrons when reacting.
- The valency of fluorine is seven. This means that the outer electron shell of a fluorine atom contains seven electrons. An easy way to determine the. See full answer below.
Comprehensive data on the chemical element Fluorine is provided on this page; including scores of properties, element names in many languages, most known nuclides of Fluorine. Common chemical compounds are also provided for many elements. In addition technical terms are linked to their definitions and the menu contains links to related articles that are a great aid in one's studies.
Fluorine Menu
- Fluorine Page One
- Fluorine Page Two
- Fluorine Page Three
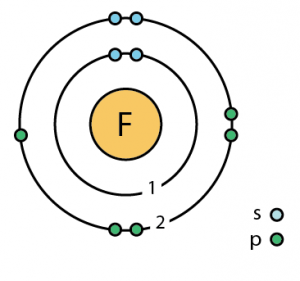
Fluorine is a halogen. All halogens have 7 valence electrons. There are thus 7 valence electrons for Fluorine, 2 in the 2s orbital, and 5 in the 2p orbitals. Most atoms achieve a stable number of valence electrons by sharing electrons with other atoms. Begin with fluorine, element 9, which has the electron configuration 1 s 2 2 s 2 2 p 5. The orbitals of the valance electron shell are 2 s 2 p, with two electrons in the 2 s and five electrons in the 2 p. These seven valence electrons can be portrayed in a diagram devised by the American chemist Gilbert Lewis (1875–1946).
Overview of Fluorine
- Atomic Number: 9
- Group: 17
- Period: 2
- Series: Halogens
Fluorine's Name in Other Languages
- Latin: Fluorum
- Czech: Fluor
- Croatian: Fluor
- French: Fluor
- German: Fluor - r
- Italian: Fluoro
- Norwegian: Fluor
- Portuguese: Flúor
- Russian: Фтор
- Spanish: Flúor
- Swedish: Fluor
Atomic Structure of Fluorine
- Atomic Radius: 0.57Å
- Atomic Volume: 17.1cm3/mol
- Covalent Radius: 0.72Å
- Cross Section (Thermal Neutron Capture)σa/barns: 0.0096
- Crystal Structure: Cubic
- Electron Configuration:
- 1s2 2s2p5
- Electrons per Energy Level: 2,7
- Shell Model
- Shell Model
- Ionic Radius: 1.33Å
- Filling Orbital: 2p5
- Number of Electrons (with no charge): 9
- Number of Neutrons (most common/stable nuclide): 10
- Number of Protons: 9
- Oxidation States: -1
- Valence Electrons: 2s2p5
- Electron Dot Model
- Electron Dot Model
Chemical Properties of Fluorine
Fluorine Valence Electrons
- Electrochemical Equivalent: 0.70883g/amp-hr
- Electron Work Function:
- Electronegativity: 3.98 (Pauling); 4.1 (Allrod Rochow)
- Heat of Fusion: 0.2552kJ/mol
- Incompatibilities:
- Water, nitric acid, oxidizers, organic compounds
- Ionization Potential
- First: 17.422
- Second: 34.97
- Third: 62.707
- Valence Electron Potential (-eV): -10.1
Fluorine Valence Sum
Physical Properties of Fluorine
- Atomic Mass Average: 18.9984
- Boiling Point: 85.1K -188.05°C -306.49°F
- Coefficient of lineal thermal expansion/K-1: N/A
- Conductivity
- Electrical:
Thermal: 0.000279 W/cmK
- Electrical:
- Density: 1.696g/L @ 273K & 1atm
- Description:
- Greenish-yellow gas of the Halogen family
- Enthalpy of Atomization: 79.08 kJ/mole @ 25°C
- Enthalpy of Fusion: 0.26 kJ/mole
- Enthalpy of Vaporization: 3.31 kJ/mole
- Flammablity Class: Non-flammable gas (extreme oxidizer)
- Freezing Point:see melting point
- Heat of Vaporization: 3.2698kJ/mol
- Melting Point: 53.63K -219.52°C -363.14°F
- Molar Volume: 17.1 cm3/mole
- Optical Refractive Index: 1.000195
- Physical State (at 20°C & 1atm): Gas
- Realitive Gas Density (Air=1) = 1.31
- Specific Heat: 0.82J/gK
Regulatory / Health
Fluorine Valence Orbitals
- CAS Number
- 7782-41-4 cryogenic liquid
- UN/NA ID and ERG Guide Number
- 1045 / 124 compressed
- 9192 / 167 cryogenic liquid
- RTECS: LM6475000
- NFPA 704
- Health:
- Fire:
- Reactivity:
- Special Hazard: OxidizerOSHAPermissible Exposure Limit (PEL)
- 1 ppm = 1.55mg/m3 @ 25°C & 1 atm
- TWA: 0.1 ppm
- OSHA PEL Vacated 1989
- TWA: 0.1 ppm
- NIOSHRecommended Exposure Limit (REL)
- TWA: 0.1 ppm
- IDLH: 25 ppm
- Routes of Exposure: Inhalation; Skin and/or eye contact
- Target Organs: Eyes, skin, respiratory system, liver, kidneys
- Levels In Humans:
Note: this data represents naturally occuring levels of elements in the typical human, it DOES NOT represent recommended daily allowances.- Blood/mg dm-3: 0.5
- Bone/p.p.m: 2000-12,000
- Liver/p.p.m: 0.22-7
- Muscle/p.p.m: 0.05
- Daily Dietary Intake: 0.3-0.5 mg
- Total Mass In Avg. 70kg human: 2.6 g
Who / Where / When / How
- Discoverer: Henri Moissan
- Discovery Location: Paris France
- Discovery Year: 1886
- Name Origin:
- Latin: fluo (flow).
- Abundance of Fluorine:
- Earth's Crust/p.p.m.: 950
- Seawater/p.p.m.:
- Atlantic Suface: 0.0001
- Atlantic Deep: 0.000096
- Pacific Surface: 0.0001
- Pacific Deep: 0.00004
- Atmosphere/p.p.m.: N/A
- Sun (Relative to H=1E12): 0.000363
- Sources of Fluorine:
- Found in the minerals fluorite (CaF2) and cryolite (Na2AlF6). Around 2,400 tons of fluorine gas and 4,700,000 tons of fluorite are produced each year. Primary mining areas are Canada, USA, UK, Russia, Mexico and Italy.
- Uses of Fluorine:
- Combines more readily than any other element. Used in refrigerants and other chloro fluorocarbons. Also in toothpaste as sodium fluoride (NaF) and stannous fluoride (SnF2); also in Teflon.
- Additional Notes:
- Fluorine gas is highly toxic and corrosive. Even exposure to low concentrations causes lung and eye irritation. Metal fluorides are also very toxic while organic fluorides are often quite harmless.
Fluorine Menu
- Fluorine Page One
- Fluorine Page Two
- Fluorine Page Three
References
A list of reference sources used to compile the data provided on our periodic table of elements can be found on the main periodic table page.
Related Resources
- Anatomy of the Atom
Answers many questions regarding the structure of atoms. - Molarity, Molality and Normality
Introduces stoichiometry and explains the differences between molarity, molality and normality. - Molar Mass Calculations and Javascript Calculator
Molar mass calculations are explained and there is a JavaScript calculator to aid calculations. - Chemical Database
This database focuses on the most common chemical compounds used in the home and industry.
Citing this page
If you need to cite this page, you can copy this text:
Kenneth Barbalace. Periodic Table of Elements - Fluorine - F. EnvironmentalChemistry.com. 1995 - 2021. Accessed on-line: 4/25/2021
https://EnvironmentalChemistry.com/yogi/periodic/F.html
.Linking to this page
If you would like to link to this page from your website, blog, etc., copy and paste this link code (in red) and modify it to suit your needs:
<a href='https://EnvironmentalChemistry.com/yogi/periodic/F.html'>echo Periodic Table of Elements: Fluorine - F (EnvironmentalChemistry.com)</a>- Comprehensive information for the element Fluorine - F is provided by this page including scores of properties, element names in many languages, most known nuclides and technical terms are linked to their definitions.
.NOTICE: While linking to articles is encouraged, OUR ARTICLES MAY NOT BE COPIED TO OR REPUBLISHED ON ANOTHER WEBSITE UNDER ANY CIRCUMSTANCES.
PLEASE, if you like an article we published simply link to it on our website do not republish it.
